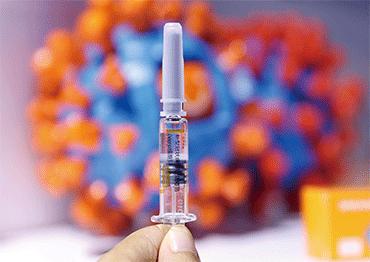
China authorized the emergency use of Covid-19 vaccines on July 22, said a senior health official in a TV interview with State broadcaster China Central Television (CCTV).
Zheng Zhongwei, director of the Development Center for Medical Science and Technology at the National Health Commission (NHC), who also heads China’s coronavirus vaccine development task force, said the emergency vaccination prioritized medical staff, epidemic prevention workers, border inspectors and those who help ensure the basic functioning of cities.
The official with China’s top health authority noted a slew of plans have been formulated to ensure proper regulation and monitoring of the emergency use.
The pricing of Covid-19 vaccines in the future will be based on their actual costs, rather than market supply, due to the vaccine’s nature as a public health product, Zheng said.
According to China’s Vaccines Administration Law, the NHC can apply for emergency use of vaccines that are still undergoing clinical trials in a public health emergency. Such use must obtain approval from an expert review organized by the State Food and Drug Administration before it can be performed within a limited scope and a limited period of time.
 Old Version
Old Version